Lapwing

Introduction
The noisy, tumbling display flights of this green and white plover are a welcome, if increasingly uncommon, sign of spring.
The Lapwing is a handsome wader with its glossy dark green plumage (often looking black from a distance) and long crest. It used to breed commonly over much of lowland Britain, but atlas projects have charted a reduction in the number of breeding locations, caused by changes in land management and increased predation pressure.
In winter, the Lapwing is widely distributed in lowland Britain, but BTO Surveys have shown that numbers have both shifted eastward (perhaps a response to milder winters) and increased in coastal wetlands as birds appear to feed more on mudflats at low tide.
- Our Trends Explorer gives you the latest insight into how this species' population is changing.

Key Stats
Identification
Songs and Calls
Song:
Alarm call:
Status and Trends
Conservation Status
Population Change
Although CBC recorded some increase in its early years, Lapwings have declined continuously on lowland farmland since the mid 1980s. National surveys in England and Wales showed a 49% population decline between 1987 and 1998 (Wilson et al. 2001). In Northern Ireland, the breeding population had shrunk to just 860 (277-1545) pairs by 2013, representing a decrease of around 89% since 1987, with the distribution becoming increasingly fragmented (Colhoun et al. 2015). Population declines there mirror similar declines throughout wet meadow areas of Wales and southeast England (Wilson et al. 2001, 2005a). The BBS map of change in relative density between 1994-96 and 2007-09 indicates that decreases have been strongest in lowland regions and the south and that some increase may have occurred in some upland and northern regions of Britain. Stability in breeding numbers in the Uists between 1983 and 2014 runs against the UK trend and heightens the relative importance of this population (Calladine et al. 2015). Winter numbers counted by WeBS, mainly at coastal sites and omitting some big concentrations inland, increased in Britain during the 1980s and early 1990s, but decreased steeply between 2005 and 2010 (Frost et al. 2020); these birds are mainly of continental origin. Lapwing is in decline across Europe, having decreased in all regions since 1980, although with differing regional timing (PECBMS: PECBMS 2009, PECBMS 2020a>). The 2009 review moved this species from amber to the UK red list, for which it continues to qualify on the strength of its UK decline.
Distribution
In Britain, wintering concentrations of Lapwings are highest in the Somerset Levels, Fens, northwest England, the Uists, Caithness and Orkney. The former core area in central England is less dominant and Fenland and the east coast now more prominent than in the 1981–84 Winter Atlas. Densities of breeding Lapwings are highest in northwest England, Orkney, Shetland and the Outer Hebrides; those in Ireland are low throughout.
Occupied 10-km squares in UK
2007/08–10/11
or view it on Bird Atlas Mapstore.
2008–11
or view it on Bird Atlas Mapstore.
European Distribution Map
Distribution Change
Despite decades of population decline, Lapwings are still the most widespread breeding waders in Britain & Ireland. Over the last 40 years, they have been lost as breeders from most of southwest England, west Wales and western mainland Scotland. Losses throughout Ireland mean that the current range is less than half that in the 1968–72 Breeding Atlas.
Change in occupied 10-km squares in the UK
from 1981–84 to 2007–11
or view it on Bird Atlas Mapstore.
from 1968–72 to 2008–11
or view it on Bird Atlas Mapstore.
Seasonality
Lapwings are widely reported and present year-round.
Weekly pattern of occurrence
The graph shows when the species is present in the UK, with taller bars indicating a higher likelihood of encountering the species in appropriate regions and habitats.
Habitats
Breeding season habitats
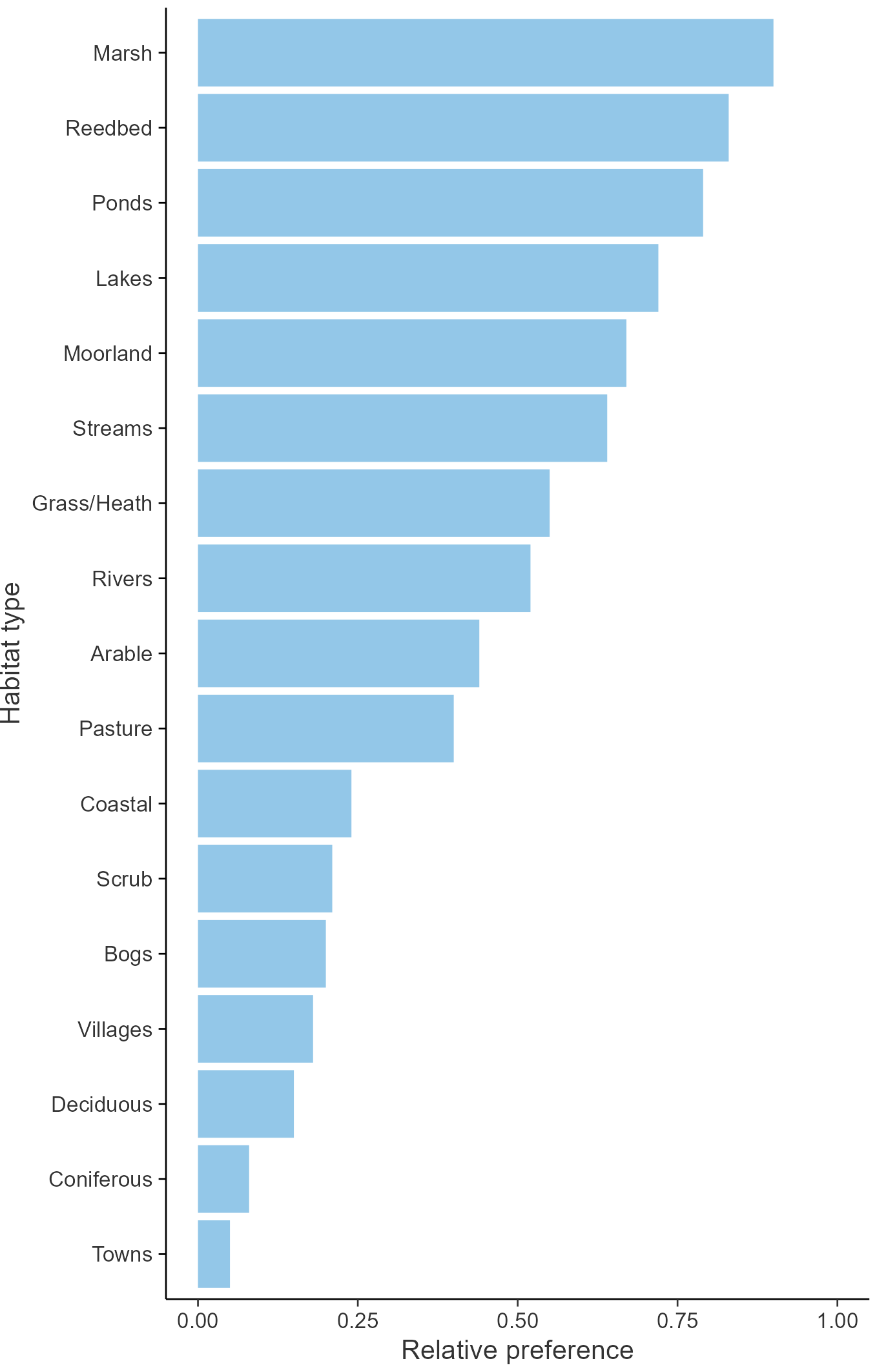
Relative frequency by habitat
The graph shows the habitats occupied in the breeding season, with the most utilised habitats shown at the top. Bars of similar size indicate the species is equally likely to be recorded in those habitats.
Movement
Britain & Ireland movement
Foreign locations of birds ringed or recovered in Britain & Ireland
Dots show the foreign destinations of birds ringed in Britain & Ireland, and the origins of birds ringed overseas that were subsequently recaptured, resighted or found dead in Britain & Ireland. Dot colours indicate the time of year that the species was present at the location.
- Winter (Nov-Feb)
- Spring (Mar-Apr)
- Summer (May-Jul)
- Autumn (Aug-Oct)

European movements
EuroBirdPortal uses birdwatcher's records, such as those logged in BirdTrack to map the flows of birds as they arrive and depart Europe. See maps for this species here.
The Eurasian-African Migration Atlas shows movements of individual birds ringed or recovered in Europe. See maps for this species here.
Biology
Productivity and Nesting
Nesting timing
Egg measurements
Clutch Size
Survival and Longevity
Survival is shown as the proportion of birds surviving from one year to the next and is derived from bird ringing data. It can also be used to estimate how long birds typically live.
View number ringed each year in the Online Ringing Report.
Lifespan
Survival of adults
Survival of juveniles
Biometrics
Wing length and body weights are from live birds (source).
Wing length
Body weight
Ring Size
Classification, names and codes
Classification and Codes
- Order: Charadriiformes
- Family: Charadriidae
- Scientific name: Vanellus vanellus
- Authority: Linnaeus, 1758
- BTO 2-letter code: L.
- BTO 5-letter code: LAPWI
- Euring code number: 4930
Alternate species names
- Catalan: fredeluga europea
- Czech: cejka chocholatá
- Danish: Vibe
- Dutch: Kievit
- Estonian: kiivitaja
- Finnish: töyhtöhyyppä
- French: Vanneau huppé
- Gaelic: Curracag
- German: Kiebitz
- Hungarian: bíbic
- Icelandic: Vepja
- Irish: Pilibín
- Italian: Pavoncella
- Latvian: kivite
- Lithuanian: paprastoji pempe
- Norwegian: Vipe
- Polish: czajka (zwyczajna)
- Portuguese: abibe
- Slovak: cíbik chochlatý
- Slovenian: priba
- Spanish: Avefría europea
- Swedish: tofsvipa
- Welsh: Cornchwiglen
- English folkname(s): Peewit, Green Plover, Hornpie, Flopwing
Research
Causes of Change and Solutions
Causes of change
There is good evidence that declines have resulted from habitat loss and degradation due to changes in agricultural practice, in particular change from spring to autumn sowing, drainage of grasslands and loss of mixed farmland, which have led to breeding productivity dropping below a sustainable level. Chick mortality is thought to be the main determinant of poor Lapwing productivity, and therefore of population decline.
Further information on causes of change
The decline of the Lapwing exemplifies how different factors can combine to cause population change (Robinson et al. 2014). The decrease in the 1980s was due to a period of low survival, when annual life expectancy decreased from 7 years to just 4.5 years, caused by a series of cold winters. At the same time, though, average nest survival decreased markedly, meaning the population could not recover from what would normally have been only a temporary setback.
There is a good deal of research supporting the hypothesis that habitat loss and degradation due to the intensification of farming have reduced breeding productivity (e.g. Galbraith 1988, Shrubb 1990, Hotker 1991, Hudson et al. 1994, Siriwardena et al. 2000a, Taylor & Grant 2004, Wilson et al. 2005, Milsom 2005, Fuller & Ausden 2008). These changes include extensive drainage, increased use of pesticides and fertilisers, re-seeding, earlier and more frequent mowing, increased grazing pressure and loss of spring cereals. Increases in intensity of grazing have reduced the habitat quality for Lapwing (Shrubb 1990, Fuller & Ausden 2008), whilst fertilisation has led to earlier spring grass growth, earlier cutting dates and higher stocking levels, which have increased egg and chick mortality and reduced relaying opportunities (Durant et al. 2008). Drainage and loss of wet features on grassland have also had a negative impact, reducing food supplies (Taylor & Grant 2004, Eglington et al. 2010).
Loss of mixed farming systems and extensive grazing have reduced the availability of high-quality foraging habitat close to nesting habitat, i.e. unimproved pasture and meadows, to birds breeding in arable areas, resulting in reduced breeding success (Galbraith 1988, Hudson et al. 1994, Henderson et al. 2004).
In the uplands, afforestation has also resulted in habitat loss (Fuller & Ausden 2008). It is possible that afforestation may also cause indirect as well as direct habitat loss: a study in Estonia found that breeding sites were preferentially located further away from woodland and suggested that woodland planting may have contributed to declines there (Kaasiku et al. 2019); however, similar analysis has not been carried out in the UK. On arable land, spring-sown cereals were once favoured nesting crops but these have been widely replaced by autumn-sown cereals, which are less suitable breeding habitats (Shrubb 1990, Shrubb et al. 1991, Mason & Macdonald 1999, Fuller & Ausden 2008). Land use changes causing a reduction in spring sward height also probably contributed to a decline on mixed farmland habitat in Scotland (Bell & Calladine 2017).
Lapwing population declines may also be explained partly by increased nest predation rates resulting from habitat changes due to agricultural intensification (Baines 1990, Liker & Szekely 1997, Jackson & Green 2000, Chamberlain & Crick 2003, Evans 2004, Jackson et al. 2004, Milsom 2005, Bolton et al. 2007, Teunissen et al. 2008, MacDonald & Bolton 2008b, Bellebaum & Bock 2009, Mason et al. 2018). Long-term nest record card analysis has shown that the proportion of nests lost to predators was substantially higher in the 1990s than in previous decades (Sharpe et al. 2008).
Recent empirical evidence suggests that levels of predation on wader nests are unsustainably high in many cases, even in some situations where breeding habitat is otherwise favourable (MacDonald & Bolton 2008a). Laidlaw et al. (2015, 2017) found that nest predation rates in wet grassland increased as the distance from patches of taller vegetation increased, and suggested that the distribution and activity of predators might be affected by the vegetation. Small mammal activity was higher in tall vegetation, particularly later in the breeding season, hence foxes in the vicinity of vegetation patches may be attracted to them and away from Lapwing nests and chicks (Laidlaw et al. 2018). In dry fields, nest predation rates were higher further from field edges (Laidlaw et al. 2017). Predation rates are also higher in areas with low Lapwing density (Laidlaw et al. 2017).
In the Uists, where the overall population is stable (Calladine et al. 2015), clutch survival is significantly lower in areas where introduced Hedgehogs Erinaceus europaeus are more abundant; however the impact of predation on local populations was unclear and more complex factors may influence trends (Calladine et al. 2017).
A Danish study found a negative correlation between annual predation rates and the numbers of breeding Lapwing, suggesting that birds nesting at higher densities may be more efficient at defending against predators (Moller et al. 2018) and hence that declines could make populations even more vulnerable if they also lead to lower nesting densities. McCallum et al. (2015, 2016) found that Lapwing density was greatest at higher elevation, but only where soils were less peaty and less acidic, opening the way to trials of whether soil amendments such as liming could contribute to conservation management for breeding Lapwings and other species of concern that depend upon soil-dwelling invertebrates. Declines among Lapwings are unlikely to be ameliorated by either habitat improvement or predator control in isolation, however (Bodey et al. 2011, Smart et al. 2013).
Information about conservation actions
As a result of the declines, the Lapwing has been the subject of a substantial amount of recent research and a number of conservation actions have been suggested. Local management actions to improve breeding habitat could include the creation of Lapwing plots in arable fields (Schmidt et al. 2017); sowing spring cereals rather than autumn cereals; delaying mowing or grazing; management of water levels to restore wetland features and habitats (Eglington et al. 2010); the exclusion of livestock from selected areas on coastal grazing marshes where the rate of grass growth is slow in spring (Hart et al. 2002); and soil amendment on upland farmland (McCallum et al. 2015).
The exclusion of predators such as foxes may also be important (see Causes of Change section), and can improve nest success (Sheldon et al. 2007; Rickenbach et al. 2011; Malpas et al. 2013b), although one study found that predator control did not affect nesting success (Bodey et al. 2011). An alternative, and possibly more sustainable, approach could be to influence predation rates by the creation and management of habitat to influence the movements of predators away from nests; for example taller vegetation patches which attract predators as a result of higher vole numbers, and wetland features to deter some predators (Laidlaw et al. 2015; 2017; 2018).
However, declines among Lapwings are unlikely to be ameliorated by either habitat improvement or predator control in isolation (Bodey et al. 2011, Smart et al. 2013 Smart et al. 2013) and hence an approach which combines different conservation actions across sites and across wider landscapes may be required. An Estonian study found that apparently suitable sites close to woodland were avoided (Kaasiku et al. 2019); hence habitat improvement should be focused on areas away from woodland and new woodlands should not be planted close to existing sites.
Publications (13)
Breeding Plover Survey for Common Standards Monitoring in Scotland, 2007: assessments of changes in numbers of breeding waders on SPAs designated for breeding Ringed Plovers Charadrius hiaticula
Author:
Published: 2008
In 2007, UK-wide surveys of breeding Ringed Plover Charadrius hiaticula and Little Ringed Plover Charadrius dubius were undertaken, the first national surveys for these species since 1984. Since then, declines had been reported in some local populations of Ringed Plover – notably in the stronghold of the Outer Hebrides where several waders have suffered greatly from egg predation by introduced Hedgehogs Erinaceus europaeus. This report provides an update on the current population sizes of Ringed Plover and other wader species in four Scottish Special Protection Areas (SPAs) designated for Ringed Plover. Population changes for feature wader species are calculated using recent previous estimates for the SPAs taken from the UK SPA Review.
01.10.08
BTO Research Reports

Trialling farmer wader counts and bioacoustics to aid agri-environment scheme evaluation
Author:
Published: 2026
The primary tool by which wader conservation is delivered in farmed landscapes is agri-environment schemes, whereby payments incentivise land managers to conduct beneficial management for species or habitats. Assessments of the effectiveness of such schemes have been based on infrequent, large-scale surveys which do not produce landscape- or intervention-specific evidence to facilitate local adaptations to the design of agri-environment schemes. This report details work carried out at 10 participating farms in the Yorkshire Dales National Park, where we trialled two methods that could help to evaluate the effectiveness of scheme measures at regional scales.
27.03.26
BTO Research Reports

Efficacy of methods for producing population trends of breeding waders from Breeding Bird Survey data
Author:
Published: 2025
This report investigates the efficacy of methods for producing population trends from BTO/JNCC/RSPB Breeding Bird Survey data for six breeding wader species in the UK. It examines the effects of increasing the count threshold on the population trends for these six species, and also explores the effects of different geographic exclusion rules on the population trend of Golden Plover. Overall, it aims to test for any potential biases in the current trends, and in doing so, determines whether we can create a better approach which provides the most robust data for wader conservation moving forwards.
23.06.25
BTO Research Reports

Landscape context influences efficacy of protected areas and agri-environment scheme delivery for breeding waders
Author:
Published: 2025
16.04.25
Papers

Watching Out for Waders: The Working for Waders Nest Camera Project
Author:
Published: 2024
This report presents the results of a trial involving the use of trail cameras by land managers and other wader conservation stakeholders to monitor the outcome of wader nesting attempts. It presents the results of the trial and assesses the potential for the project to improve wader conservation knowledge and management.
04.10.24
BTO Research Reports

Loss of breeding waders from key lowland grassland sites in Northern Ireland
Author:
Published: 2022
Between the mid-1980s and 2018–2019, Northern Ireland’s lowland wet grasslands saw a 73% decline in their breeding wader populations, from 1,296 to 354 pairs across 74 surveyed sites.
18.07.22
Papers

Nesting dates of Moorland Birds in the English, Welsh and Scottish Uplands
Author:
Published: 2022
Rotational burning of vegetation is a common form of land management in UK upland habitats, and is restricted to the colder half of the year, with the time period during which burning may be carried out in upland areas varying between countries. In England and Scotland, this period runs from the 1st October to 15th April, but in the latter jurisdiction, permission can be granted to extend the burning season to 30th April. In Wales, this period runs from 1st October to 31st March. This report sets out timing of breeding information for upland birds in England, Scotland and Wales, to assess whether rotational burning poses a threat to populations of these species, and the extent to which any such threat varies in space and time.
17.02.22
BTO Research Reports

Sensitivity mapping for breeding waders in Britain: towards producing zonal maps to guide wader conservation, forest expansion and other land-use changes. Report with specific data for Northumberland and north-east Cumbria
Author:
Published: 2021
Breeding waders in Britain are high profile species of conservation concern because of their declining populations and the international significance of some of their populations. Forest expansion is one of the most important, ongoing and large-scale changes in land use that can provide conservation and wider environmental benefits, but also adversely affect populations of breeding waders. We describe models to be used towards the development of tools to guide, inform and minimise conflict between wader conservation and forest expansion. Extensive data on breeding wader occurrence is typically available at spatial scales that are too coarse to best inform waderconservation and forestry stakeholders. Using statistical models (random forest regression trees) we model the predicted relative abundances of 10 species of breeding wader across Britain at 1-km square resolution. Bird data are taken from Bird Atlas 2007–11, which was a joint project between BTO, BirdWatch Ireland and the Scottish Ornithologists’ Club, and modelled with a range of environmental data sets.
09.12.21
BTO Research Reports

Monitoring breeding waders in Wensleydale: trialling surveys carried out by farmers and gamekeepers
Author:
Published: 2017
This report details pilot work in Wensleydale to test methods for involving farmers and gamekeepers in the survey and monitoring of breeding waders, and consider how such methods could be applied more widely.
21.11.17
BTO Research Reports

Consequences of population change for local abundance and site occupancy of wintering waterbirds
Author:
Published: 2017
Protected sites for birds are typically designated based on the site’s importance for the species that use it. For example, sites may be selected as Special Protection Areas (under the European Union Directive on the Conservation of Wild Birds) if they support more than 1% of a given national or international population of a species or an assemblage of over 20,000 waterbirds or seabirds. However, through the impacts of changing climates, habitat loss and invasive species, the way species use sites may change. As populations increase, abundance at existing sites may go up or new sites may be colonized. Similarly, as populations decrease, abundance at occupied sites may go down, or some sites may be abandoned. Determining how bird populations are spread across protected sites, and how changes in populations may affect this, is essential to making sure that they remain protected in the future. These findings come from a new study by Verónica Méndez and colleagues from the University of East Anglia working with BTO. Using Wetland Bird Survey (WeBS) data the study looked at changes in the population sizes and distributions of 19 waterbird species across Britain during a period of 26 years and their effect on local abundance and site occupancy. Some of these species saw steady increases in population size (up to 1,600%, Avocet), whereas other saw mild declines (-26%, Purple Sandpiper and Shelduck). The results showed that changes in total population size were predominantly reflected in changes in local abundance, rather than through the addition or loss of sites. This is possibly because waterbirds tend to be long-lived birds, with high site fidelity and new suitable sites may not always be available. Thus colonisation of new sites may typically occur when their existing sites approach their maximum capacity. As changes in populations are largely manifested by changes in local abundance – and as sites are often designated for many species – the numbers of sites qualifying for site designation are unlikely to be affected. Understanding the dynamic between population change and change in local abundance will be key to ensuring the efficiency of protected area management and ensuring that populations are adequately protected. Data from the Wetland Bird Survey and its predecessor schemes, which are celebrating 70 years of continuous monitoring of waterbirds this year, have been integral to both the designation of protected sites and monitoring of their condition. Continuation of this monitoring through future generations will ensure that the impacts to waterbird populations of future environmental changes may be understood.
20.09.17
Papers

The decline of a population of farmland breeding waders: a twenty-five-year case study
Author:
Published: 2017
The breeding populations of many different wader species are in decline across the globe, and the UK is no exception. These declines have been linked to increased predator numbers, changes in agricultural practices, and in the management of the wider landscape. There is an urgent need for information on how such changes in land management, particularly within farmland, may affect breeding waders. This information can then be used to inform future land management decisions. Long-term studies can make an important contribution to our understanding of wader decline, as is demonstrated by work carried out by BTO’s Regional representative for Perthshire, Mike Bell. Mike had the foresight to start monitoring what was an important concentration of breeding waders in Strathallan (Perthshire, Scotland) in the late 1980s. Twenty-five years after establishing repeatable methods over the study area proved an appropriate time to take stock of changes that occurred. The four most numerous species in the area were Oystercatcher, Lapwing, Curlew and Redshank, which respectively achieved densities of 12, 36, 3 and 5 pairs per km2 in 1990. All are facing serious declines across the UK. Alongside information on the breeding populations of these four waders, Mike also documented agricultural changes; from this it was possible to determine how local farming practices were likely to have influenced the waders. Over the period of 1990-2015 Oystercatchers showed the greatest proportional decline with the breeding population falling by 95%, followed by Lapwing (-88%), Redshank (-87%) and Curlew (-67%). Although the greatest declines were associated with changing land use, the declines appeared greater than could be accounted by losses of preferred habitat alone. Furthermore, breeding success was low throughout the study and probably not sufficient to maintain the population. This raises the importance of understanding the roles of source and sink populations for breeding waders in the wider countryside. Alongside this, threats from poor weather, increased disturbance (e.g. from dog walkers), and increased predator numbers could further implicate the population, although this was not studied specifically at Strathallan. Mike Bell is a long-term volunteer for the BTO and now Regional Representative for Perthshire. This 25-year (and ongoing) study was undertaken in his own time, highlighting the value of volunteers to science and conservation. It also emphasises how long-term datasets such as this can provide invaluable insights into the pressures that bird species are facing, and how we may alleviate these pressures. As most long-term studies are possible only with volunteers, the importance of their contribution is increasingly important. With support from the BTO’s recent Curlew Appeal, BTO has been trialling a range of approaches where volunteers can add to our understanding of the ecology of breeding waders, knowledge which will be vital to provide these beautiful birds with a fighting chance.
12.04.17
Papers Bird Study

Distribution shifts in wintering Golden Plover Pluvialis apricaria and Lapwing Vanellus vanellus in Britain
Author:
Published: 2006
01.01.06
Papers Bird Study
Breeding seasons of some British waders
Author:
Published: 1982
This report summarises the findings of an analysis of BTO Nest Record Scheme cards for eight species of wader, providing data on their breeding seasons.
01.11.82
BTO Research Reports
More Evidence
More evidence from Conservation Evidence.com
Partners
Citing BirdFacts
If you wish to cite particular content in this page (e.g. a specific value) it is best to use the original sources as linked in the page. For a more general citation of the whole page please use: BTO (20XX) BirdFacts Species: profiles of birds occurring in the United Kingdom. BTO, Thetford (www.bto.org/birdfacts, accessed on xx/xx/xxxx).