Nightjar

Introduction
This summer visitor, highly cryptically coloured, is more likely to be heard than seen by visitors to its breeding sites, mostly scattered across the southern half of Britain.
Nightjars were once much more widely distributed across Britain than is the case now, occupying lowland heathland and other sites that have been lost to agriculture and afforestation. The very recent increases in numbers and range have been facilitated in places by the availability of recently-felled plantation woodland and climatic changes.
Tracking studies have confirmed that the main wintering area is located in the scrub dominated grasslands, primarily within the Democratic Republic of Congo, that sit to the south of the equatorial rainforest.
- Our Trends Explorer gives you the latest insight into how this species' population is changing.

Key Stats
Identification
Songs and Calls
Song:
Call:
Alarm call:
Flight call:
Status and Trends
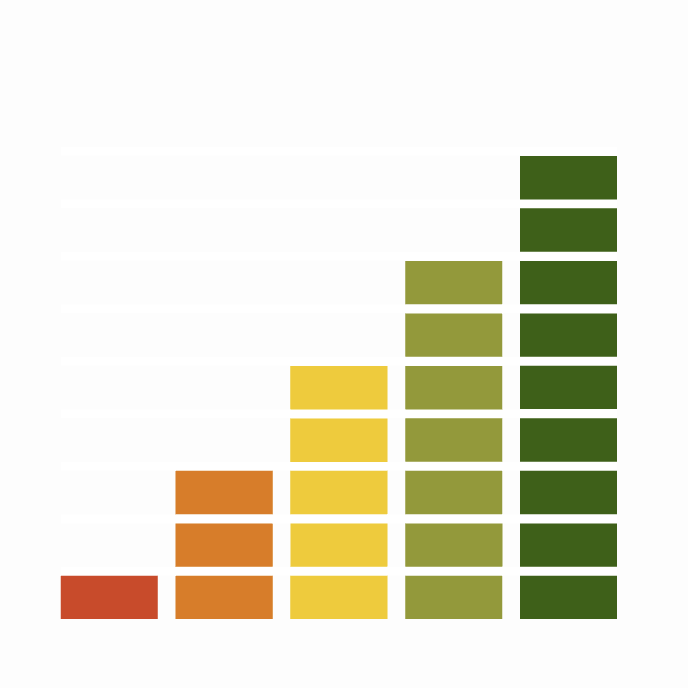
Conservation Status
Population Change
Following a catastrophic decline in range of more than 50% of 10-km squares between the 1968-72 and 1988-91 breeding atlases, the 1992 national survey revealed a welcome increase of 50% in population size since an earlier survey in 1981 (Morris et al. 1994). A national Nightjar Survey in 2004 revealed that a further 36% increase had taken place in the UK population in 12 years, with a 2.6% increase in the number of 10-km squares occupied (Conway et al. 2007). There was evidence of population declines and range contractions since 1992, however, in North Wales, northwest England, and Scotland. Atlas data from 2008-11 show an 18% range increase in Britain since 1988-91 but some parts of the 1968-72 range remain unoccupied (Balmer et al. 2013). Through its partial recovery of UK range, the species moved from red to being amber listed in the latest review (Eaton et al. 2015).
Although annual nest record samples are very small, nest failure rates have increased and clutch size has decreased slightly. A steep decrease was evident until the early 2000s in the number of fledglings per breeding attempt; a slight improvement in the last ten years has not yet reversed the earlier decrease.
Distribution
Across much of its range, the Nightjar's the breeding distribution is closely associated with lowland heathland and felled or recently planted conifer plantations, though coastal moorland, Sweet Chestnut coppice and sand dunes may also be occupied. In Ireland they mainly occupy clear-felled conifer plantations.
Occupied 10-km squares in UK
2008–11
or view it on Bird Atlas Mapstore.
European Distribution Map
Distribution Change
Historically, Nightjars were widely distributed throughout Britain & Ireland but between the 1968–72 Breeding Atlas and the 1988–91 Breeding Atlas the range contracted by 51% in Britain and by 88% in Ireland. More recently, an 18% range expansion in Britain from 1988–91 to 2008–11 sees gains beyond the previous distribution and includes a novel broadening of habitat use whereby moorland conifer plantations are now being occupied; this may assist future range expansion.
Change in occupied 10-km squares in the UK
from 1968–72 to 2008–11
or view it on Bird Atlas Mapstore.
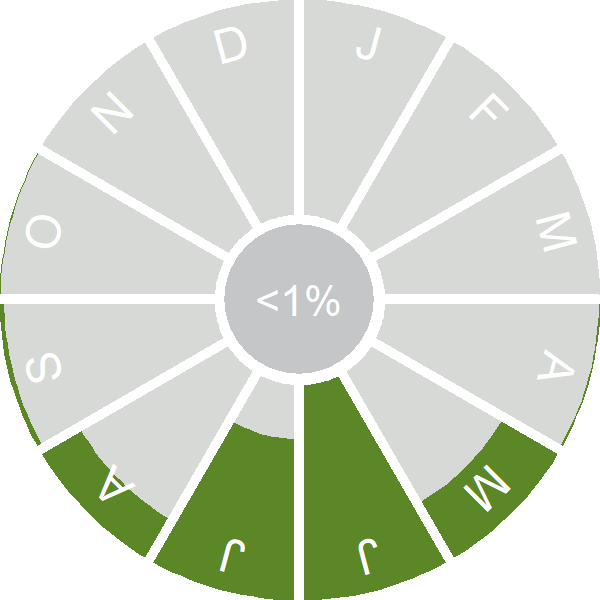
Seasonality
Nightjars are summer visitors, present from May to mid August.
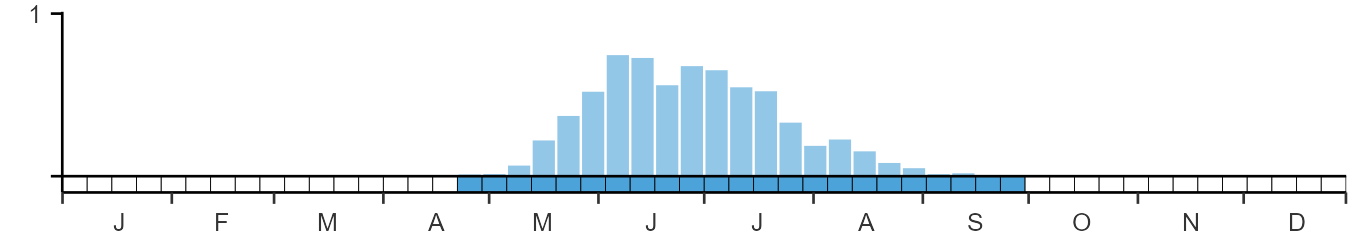
Weekly pattern of occurrence
The graph shows when the species is present in the UK, with taller bars indicating a higher likelihood of encountering the species in appropriate regions and habitats.
Movement
Britain & Ireland movement
Foreign locations of birds ringed or recovered in Britain & Ireland
Dots show the foreign destinations of birds ringed in Britain & Ireland, and the origins of birds ringed overseas that were subsequently recaptured, resighted or found dead in Britain & Ireland. Dot colours indicate the time of year that the species was present at the location.
- Winter (Nov-Feb)
- Spring (Mar-Apr)
- Summer (May-Jul)
- Autumn (Aug-Oct)

European movements
EuroBirdPortal uses birdwatcher's records, such as those logged in BirdTrack to map the flows of birds as they arrive and depart Europe. See maps for this species here.
The Eurasian-African Migration Atlas shows movements of individual birds ringed or recovered in Europe. See maps for this species here.
Biology
Productivity and Nesting
Nesting timing
Egg measurements
Clutch Size
Incubation
Fledging
Survival and Longevity
Survival is shown as the proportion of birds surviving from one year to the next and is derived from bird ringing data. It can also be used to estimate how long birds typically live.
View number ringed each year in the Online Ringing Report.
Lifespan
Survival of adults
Biometrics
Wing length and body weights are from live birds (source).
Wing length
Body weight
Ring Size
Classification, names and codes
Classification and Codes
- Order: Caprimulgiformes
- Family: Caprimulgidae
- Scientific name: Caprimulgus europaeus
- Authority: Linnaeus, 1758
- BTO 2-letter code: NJ
- BTO 5-letter code: NIJAR
- Euring code number: 7780
Alternate species names
- Catalan: enganyapastors comú
- Czech: lelek lesní
- Danish: Natravn
- Dutch: Nachtzwaluw
- Estonian: öösorr
- Finnish: kehrääjä
- French: Engoulevent d’Europe
- Gaelic: Seabhag-oidhche
- German: Ziegenmelker
- Hungarian: lappantyú
- Icelandic: Náttfari
- Irish: Tuirne Lín
- Italian: Succiacapre
- Latvian: vakarlepis
- Lithuanian: europinis lelys
- Norwegian: Nattravn
- Polish: lelek (zwyczajny)
- Portuguese: noitibó-cinzento
- Slovak: lelek lesný
- Slovenian: podhujka
- Spanish: Chotacabras europeo
- Swedish: nattskärra
- Welsh: Troellwr Mawr
- English folkname(s): Dor-hawk, Fern Owl
Research
Causes of Change and Solutions
Causes of change
The recovery of this species coincided with the availability of suitable open ground habitat resulting from the felling of forests planted in the late 1920s and 1930s, the clearance and restocking of areas damaged by storms in the late 1980s and, importantly, the restoration of heathland habitats. Management, protection, restoration and re-creation of key habitats remains critical for maintaining Nightjar numbers.
Further information on causes of change
The historical population decline and contraction of range have been attributed to large-scale losses of heathland to agriculture, construction and afforestation (Conway et al. 2007, Langston et al. 2007b). Recovery has coincided with more suitable open ground becoming available through the felling of forests planted in the late 1920s and 1930s, the clearance and restocking of areas damaged by storms in 1987 and 1990 and the restoration of heathland (Scott et al. 1998, Ravenscroft 1989, Morris et al. 1994, Conway et al. 2007, Langston et al. 2007b). While most recent increase has been consolidation within the existing range, there has been colonisation of conifer plantations at higher altitude in southwest England and on the North York Moors: this might be a density-dependent effect as new habitat becomes available or could be evidence of positive effects of climate change (G.J. Conway pers comm).
Prospects for further recovery may be limited, however, due to a reduction of suitable habitat as newly restocked forests grow and to the effects of human disturbance: studies have found that concentrated human disturbance can affect territory densities (Liley & Clarke 2003; Pouwels et al. 2020) and that nest failure is most likely in areas heavily frequented by walkers and dogs (Langston et al. 2007a). However another study, in Thetford Forest, concluded that recreational disturbance was not a factor in nest failure (Dolman 2010), and a study in Nottinghamshire found that, although territory selection was influenced by disturbance, there appeared to be no concurrent impact on breeding success (Lowe et al. 2014). The Thetford study also observed that all nest predators were mammalian (foxes and badgers), but their impact was unlikely to affect Nightjar population size (Dolman 2010). In Switzerland, a study concluded that denser vegetation regrowth and lower prey abundance hindered reuse of previously occupied sites (Winiger et al. 2018); however this was directly contradicted by a subsequent study in the same area which concluded instead that prey abundance had not changed and that recolonization of apparently suitable sites was prevented by light pollution (Sierro & Erhardt 2019).
Burgess et al. (1990) reported that, at Minsmere, creating more edge habitat and providing abundant nest sites resulted in an increase in the Nightjar population (see also Conservation Actions section, below).
New tracking studies suggest that Nightjars consistently forage in non-forest habitats, such as grasslands and semi-natural habitats, sometimes on farmlands, and that the availability and management of the adjacent landscape could affect Nightjar populations (Evens et al. 2017, 2018, Henderson/Conway, in prep.). A small-scale test study in Thetford Forest found that non-forest habitats have higher moth biomass, which Nightjars exploit, although further structured surveys are needed (Henderson et al. 2017). Tracking also suggests that some individual Nightjars may specialise by foraging within one particular habitat type whilst others follow a more generalist approach, and that habitat heterogeneity may help increase local populations by benefiting both specialists and generalists (Mitchell et al. 2020).
Information about conservation actions
Management, protection, restoration and creation (or re-creation) of key forest and heathland breeding habitats remain critical for the long-term conservation of Nightjar (Ravenscroft 1989; Morris et al. 1994; Scott et al. 1998; Conway et al. 2007). Open spaces including clear-felled areas and young stands within larger forests are important, and wide forest tracks can create additional habitat for Nightjars (Verstraeten et al. 2011). Forest management should include rotational cutting to ensure a mosaic of different aged stands are available, as well as actions such as creating glades in woodland and sculpting woodland margins to increase the area of edge habitat, leaving woodland shelter belts standing and providing abundant potential nesting sites, mainly by clearing small patches of heather from the base of small birch trees, (Burgess et al. 1990). In Thetford Forest, Dolman & Morrison (2012) found that density of Nightjars was highest in areas of restock at pre-thicket stages (6-10 years) and that management of conifer plantations plays an important role in determining the population of Nightjars. Radio-tracking there indicated that a variety of growth stages is important for this species and that grazing of open habitats within and adjacent to the forest will also be of benefit (Sharps, K. et al. 2015). Areas of heath and bare sand should also be created or maintained within the open patches (Verstraeten et al. 2011).
As discussed in the Causes of Change sections, both disturbance and light pollution may possibly affect Nightjar density and therefore actions to reduce them may benefit the species, although further research is needed to confirm that these factors do impact on the population. Distance to car park, distance to road and openness all influence visitor density, and therefore changing the location of car parks can be an effective tool to manage visitor numbers (Pouwels et al. 2020)
Recent tracking studies suggest that Nightjars also forage outside forests in grassland and other semi-natural habitats including farmland (Evens et al. 2017; 2018; Henderson/Conway in prep), so the habitat requirements are not necessarily limited to the woodland area in which the birds are nesting, and a wider landscape scale approach may be needed in addition to local site management.
Publications (7)
Impact of future climate change and land-use change on habitat suitability for a long-distance avian migrant under diverse socioeconomic-emission scenarios
Author:
Published: 2025
During the current period of accelerating change in both climate and environmental conditions many birds species are having to adapt to new conditions. While sedentary species only have to adapt to changes in one location, migrants have to contend with multiple changes throughout the annual cycle.
05.08.25
Papers

Environmental and Geographic Conditions on the Breeding Grounds Drive Bergmannian Clines in Nightjars
Author:
Published: 2025
Bergmann’s rule (or ‘law’) is one of the oldest in ecology and evolutionary biology, first presented in 1847. This rule states that animals inhabiting colder climates/latitudes are larger, with greater volume than individuals of the same species occupying warmer climates/latitudes.
12.07.25
Papers

The genomic signature of demographic decline in a long-distance migrant in a range-extreme population
Author:
Published: 2025
With increasing sophistication, genetic techniques and analyses are allowing us to delve deep into the past to show how prior environmental or demographic change has influenced species’ present day populations. This is particularly useful for scarce species that are difficult to observe and occupy remote habitats. In many species, large scale reduction in habitat availability has impacted their breeding distribution, which has been exacerbated by habitat fragmentation. Once widespread species become localised and the remaining breeding birds become increasingly isolated, this can result in reduced genetic diversity. This is the case for the Nightjar, a little-studied species, which has specialised habitat requirements and is located at the western edge of the European range.
05.06.25
Papers

Multi-scale habitat selection throughout the annual cycle of a long-distance avian migrant
Author:
Published: 2023
Long distance migrants, such as those breeding in western Europe and wintering south of the Sahara in Africa, face perilous journeys during autumn and spring. During the annual cycle, the habitats they use vary considerably between breeding and wintering sites, as well as on essential migration stopovers in very different landscapes on different continents. There are several geographical barriers to negotiate and large expanses of inhospitable habitat, such as sea and desert, to cross. In this collaborative study, high resolution GPS tracking data were used to investigate how the Nightjar achieves this journey, and the strategy the birds use to locate essential foraging habitat along the way. Small to medium sized migratory birds cannot store enough fat reserves to complete the journey in a single movement. Instead, they must stop to refuel at various stages to successfully complete each subsequent leg of the journey using a series of intermediate locations as ‘stepping stones’. This can be problematic for these migrants, as they first need to find suitable foraging habitat and feed sufficiently to get over the next barrier. Aerial insectivores have the added advantage that they can visually select good looking habits and feed opportunistically en route, but Nightjars can only do this when active during the hours of darkness. The results of this study showed, unsurprisingly, that Nightjars rapidly cross barriers and inhospitable habitats, including Mediterranean Sea, Sahara Desert and equatorial forests. Either side of these barriers, Nightjars generally slowed their migration and spent more time in mixed habits, where they could then more intensively search out locations providing foraging hotspots. In both spring and autumn, around nine separate stops were made en route, each ranging between one and 27 days in duration. This study demonstrates how Nightjars optimise their migratory flights and search for prime stopover sites by selecting areas with very diverse habitat composition, which by their nature support prey hotspots. The birds can then home in upon these with limited effort. It is still not known how Nightjars assess the habitat, but it is likely that they can visually assess the structure in the limited nighttime light, or during better illuminated twilight periods.
18.10.23
Papers

Flight Lines: Tracking the wonders of bird migration
Author:
Published: 2017
This stunning new book brings together the latest research findings, delivered through an accessible and engaging narrative by the BTO's Mike Toms, with the wonderful artwork generated through the BTO/SWLA Flight Lines project. If you have an interest in our summer migrants, then you'll welcome this fantastic opportunity to discover their stories through art and the written word. By pairing artists, storytellers and photojournalists with the researchers and volunteers studying our summer migrants, we are able to tell the stories of our migrant birds, and the work being done to secure a future for them. Includes artwork by SWLA member artists Carry Akroyd, Kim Atkinson, Federico Gemma, Richard Johnson, Szabolcs Kokay, Harriet Mead, Bruce Pearson, Greg Poole, Dafila Scott, Jane Smith, John Threlfall, Esther Tyson, Matt Underwood, Michael Warren, Darren Woodhead and others.
21.08.17
Books and guides Book

Migratory pathways, stopover zones and wintering destinations of Western European Nightjars Caprimulgus europaeus
Author:
Published: 2017
New research involving the BTO has revealed important information about the migration routes and wintering grounds of Nightjar, a species of conservation concern in the UK. Up until very recently our knowledge of Nightjar wintering areas in Africa was almost entirely based on visual observations and just two recoveries from all European ringing activities. Our knowledge of migration was slightly better thanks to the moderate number of ringing recoveries, but these only cover within Europe and a few sites in North Africa. New tracking technology is now vastly improving our understanding of Nightjars' movements, with implications for this species' conservation. The advent of geolocators allowed Nightjars to be tracked for the first time in 2009. These give daily locations accurate to around 100 km, meaning they are more than appropriate for tracking migration routes and wintering area at the continental scale. More recently, GPS devices have become sufficiently small to use, and these provide high precision locations but only once every 10 to 20 days. Using a combination of geolocators and GPS tags, researchers from Hasselt University, Biotrack, and the University of Sorbonne worked with Greg Conway and Ian Henderson from the BTO to examine the migration routes and wintering areas of 11 adult Nightjars marked at breeding sites in the UK, France and Belgium between 2009 and 2015. The results show that the main wintering area is located in the Savannah and scrub forests, to the south of the central African tropical Rainforests, mainly in the southern region of the Democratic Republic of Congo (similar to that favoured by our satellite-tagged Cuckoos). This area has not previously been identified as a wintering area for this species, but now appears to be a very important for Nightjars breeding in western Europe. The study also identified key stopover sites for the first time, on the boundaries of major ecological barriers including the Mediterranean Sea, Sahara Desert and Central African Tropical Rainforest, avoidance of the latter being somewhat surprising given the high insect resource potentially available in such habitat. Major differences in spring and autumn migration were noted, with autumn passage being on a broad front involving a fairly direct crossing of the central Saharan region, while birds in spring took a substantially longer western route back to their breeding grounds, involving a detour via West Africa of around 2000 km. Further work is underway to investigate migration routes and wintering area used by other breeding populations throughout Europe. In the UK, the Nightjar has recently been moved from the Birds of Conservation Concern Red List to the Amber List. By identifying key migration routes and wintering grounds, this study provides important information that will further aid monitoring and conservation efforts of this charismatic species.
15.02.17
Papers

Home-range size and habitat use of European Nightjars Caprimulgus europaeus nesting in a complex plantation-forest landscape
Author:
Published: 2015
Nightjar populations have increased in recent years, but some declines have been noted at sites supporting nationally important breeding populations. BTO research is using tracking technology to better understand the habitat requirements of this cryptic yet charismatic species, so that habitat can be optimally managed to conserve them. Population changes Although the British Nightjar population is generally faring well, increasing by 36% between 1992 and 2004 (Conway et al. 2007), there have been declines at some sites supporting regionally and nationally important numbers of this species. These declines have been attributed to the lack of available breeding habitat, but other factors may also be important, including disturbance through recreational activities and suboptimal habitat management. Habitat requirements During the breeding season, nesting habitat typically includes heathland and young conifer plantation. As both these habitats are dynamic, they require appropriate management to either maintain suitability for Nightjars, or to create replacement habitat when formerly suitable areas become overgrown. This can be achieved through felling and replanting in forest systems. However, foraging habitat is also essential for Nightjars and relatively little is known about which habitat types are the most important. It is also crucial to understand how close foraging and breeding habitats have to be for Nightjar management to be effective. Ultimately, land managers need appropriate habitat management guidance to ensure that the number of Nightjars that can be supported on a particular site is maximised, particularly where numbers have decreased. Use of technology It is difficult to observe nocturnal Nightjar activity, especially as birds may rapidly move substantial distances. A newly-published BTO study used VHF radio tracking devices to overcome this, following 31 individuals in Thetford Forest, East Anglia during 2009 and 2010 (Sharps et al. 2015). The results showed that conifer plantations aged 5-10 years were an important component of all territories, while newly planted forest (0-4 years) and grazed grassland were also used by Nightjars; all potentially important for foraging. Nightjars travelled a mean maximum distance of 747 m between breeding and feeding locations each night, demonstrating the importance of having foraging and nesting habitats in close proximity. Future research Following rapid advances in technology, GPS devices are now available, which can collect substantial quantities of high precision data. In 2014, 15 breeding Nightjars were successfully tracked with GPS in Thetford Forest and at Dersingham Bog National Nature Reserve, providing an objective and highly detailed insight into their movements. Initial analyses indicate that rapid foraging flights, often exceeding 3 km, are undertaken at dusk and dawn, to reach discrete locations that are re-visited on subsequent nights. During 2015, these apparent foraging hot spots will be further investigated with visits to observe actual Nightjar activity and examine prey abundance. Understanding the importance of these primary feeding locations and how they are used, will provide further improvements to landscape management operations and great benefits to Nightjar conservation.
01.01.15
Papers

More Evidence
More evidence from Conservation Evidence.com
Partners
Citing BirdFacts
If you wish to cite particular content in this page (e.g. a specific value) it is best to use the original sources as linked in the page. For a more general citation of the whole page please use: BTO (20XX) BirdFacts Species: profiles of birds occurring in the United Kingdom. BTO, Thetford (www.bto.org/birdfacts, accessed on xx/xx/xxxx).